Water Scale Remover


Water Scale Remover
The scale remover uses electrolysis process to remove scale forming components from the hard water and thus converting it into soft water.
Process of Electrolysis:
The key process of electrolysis is the interchange of atoms and ions by the removal or addition of electrons from the external circuit. The desired products of electrolysis are often in a different physical state from the electrolyte and can be removed by some physical processes. For example, in the electrolysis of Brineto produce hydrogen and chlorine, the products are gaseous. These gaseous products bubble from the electrolyte and are collected.
2 NaCl + 2 H2O2 NaOH + H2+ Cl2
A liquid containing electrolyte is produced by:
Solvation or reaction of an ionic compound with a solvent (such as water) to produce mobile ions.
An ionic compound is melted by heating.
An electrical applied potential across a pair of electrodes immersed in the electrolyte.
Each electrode attracts ions that are of the opposite charge. Positively charged ions (cations) move towards the electron providing (negative) cathode. Negatively charged ions (anions) move towards the electron-extracting (positive) anode.
In this process electrons are either absorbed or released. Neutral atoms gain or lose electrons and become charged ions that then pass into the electrolyte. The formation of uncharged atoms from ions is called discharging. When an ion gains or loses enough electrons to become uncharged (neutral) atoms, the newly formed atoms separate from the electrolyte. Positive metal ions like Cu2+deposit onto the cathode in a layer. The terms for this are electroplating, electro winning, and electro refining. When an ion gains or loses electrons without becoming neutral, its electronic charge is altered in the process. In chemistry, the loss of electrons is called oxidation, while electron gain is called reduction.
Oxidation and reduction at the electrodes.
Oxidation of ions or neutral molecules occurs in the anode. For example, it is possible to oxidize ferrous ions to ferric ions at the anode:
Fe2+(aq)Fe3+ (aq) + e-
Reduction of ions or neutral molecules occurs at the cathode. It is possible to reduce ferricyanide ions to ferrocyanide ions at the cathode:
Fe(CN)3-6+e-→ Fe(CN)4-6
Neutral molecules can also react at either of the electrodes.
For example: Benzoquinone can be reduced to hydroquinone at the cathode.
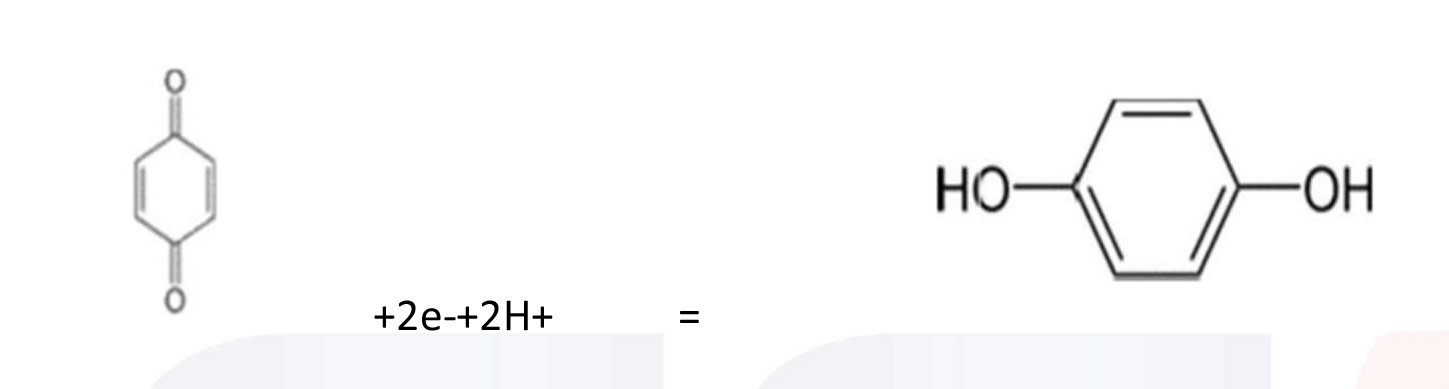
In the last example, H+ ions (hydrogen ions) also take part in the reaction, and are provided by an acid in the solution, or by the solvent itself (water, methanol etc.). Electrolysis reactions involving H+ ions are fairly common in acidic solutions. In aqueous alkaline solutions, reactions involving OH-(hydroxide ions) are common.
Sometimes the solvents themselves (usually water) are oxidized or reduced at the electrodes. It is even possible to have electrolysis involving gases. Such as when using a Gas diffusion electrode.
